Featured Posts

How Data Sharing and AI Can Reduce the Global Food Safety Burden
The following talk was recorded at the Dubai International Food Safety Conference. Sharing public-private data for food safety.

The Seven key benefits of sharing data in the realm of food safety in discussion with the FDA
The recent FDA podcast on “Artificial Intelligence in the New Era of Smarter Food Safety” highlights several key benefits of sharing data in the realm of food safety.

What are the Proposition 65 regulations?
Prop 65 is a California law that requires a clear and reasonable warning on product labels and product web pages if products contains chemicals known to cause cancer and birth defects.
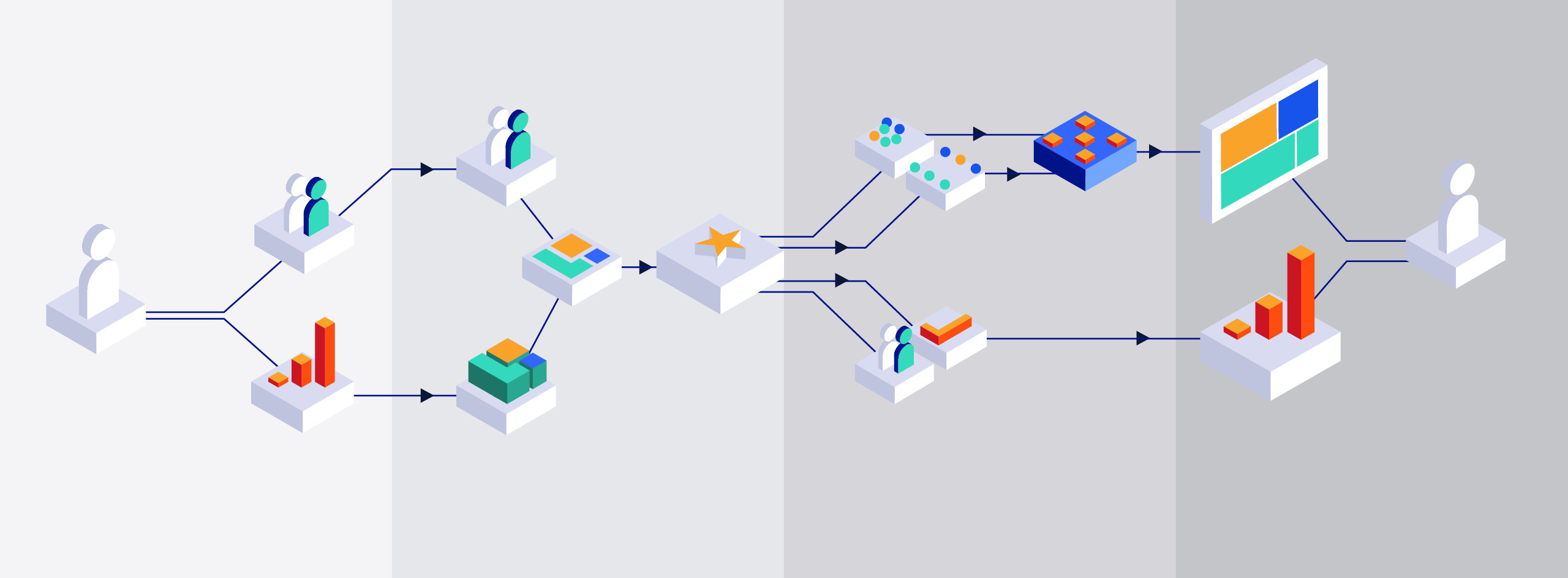
What is a Data Trust? The complete guide for organizations, regulators and manufacturers.
Data trusts provide opportunities for the members to benefit from the aggregated data, often through new data insights or predictions which can motivate data sharing.

Working with regulators and industry to reduce the salt intake of a population
Over the next two and a half years, the FDA aims to cut the average sodium level intake by 12% — from 3,400 to 3,000 milligrams a day.

Fibre fortification could lower the risk of heart disease and diabetes for 7 in 10 UK adults
Increased fibre fortification of everyday UK foods including baked goods, dairy products, soups, smoothies and dressings, will enable 50% more adults to get the recommended daily amount of fibre
Blog

The Top Cosmetic science, Fragrance and Personal Care conferences to attend
Our hand picked comprehensive list of important cosmetic industry conferences and events to attend.

The Top Food science, Nutrition and Food safety conferences to attend
Our hand picked comprehensive list of important food industry conferences and events to attend.

Thank you to everyone who joined us at SOT
A collection of photos and impressions from RIFM and Creme Global reception held at SOT 2024 conference.

Navigating Uncertainty in Food Safety with Creme Global’s Data Science and Scientific Expertise
Reflections by Creme Global CEO Cronan McNamara on his talk at the Dubai International Food Safety Conference.

How Data Sharing and AI Can Reduce the Global Food Safety Burden
The following talk was recorded at the Dubai International Food Safety Conference. Sharing public-private data for food safety.

The journey from data sharing to food safety using data trusts.
The following talk was recorded at the Dubai International Food Safety Conference. Sharing public-private data for food safety.

The Seven key benefits of sharing data in the realm of food safety in discussion with the FDA
The recent FDA podcast on “Artificial Intelligence in the New Era of Smarter Food Safety” highlights several key benefits of sharing data in the realm of food safety.

How organizations can help industries and members forecast risks and opportunities.
Embracing uncertainty – transitioning from trend forecasts to future scenarios – How data and AI can lead the way

What are the Proposition 65 regulations?
Prop 65 is a California law that requires a clear and reasonable warning on product labels and product web pages if products contains chemicals known to cause cancer and birth defects.